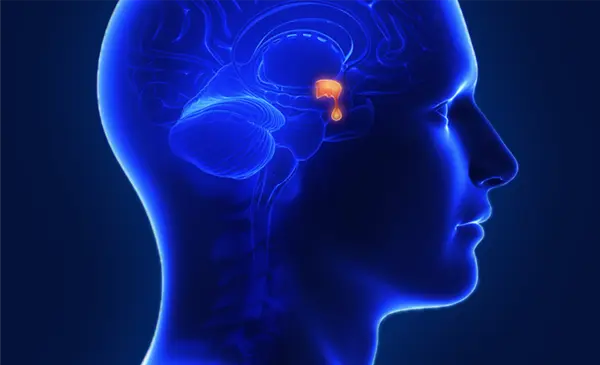
PREF-NET Study Overview: Patient Preference and Experience Study
The PREF-NET study was a patient preference and experience study that demonstrated a strong preference among patients for home administration of Lanreotide Ipsen over hospital administration. For a detailed overview, explore the study summary.
LAN-UK-000611 | June 2025